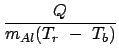You might expect to find Lv, of the nitrogen by setting
We can avoid this difficulty by noting that QAl is also the heat needed to warm the same aluminum cylinder to from Tb to Tr. You can measure this heat by placing the cold aluminum cylinder (at temperature Tb) in a ``calorimeter'' that contains water and
observing the change in temperature of the water, - ![]() T,
-provided that the final temperature of the water, Tf, is room temperature, Tr. It is hard to arrange for Tf to end up exactly at room temperature, but if Tf is close to Tr, one can accurately correct the calorimeter data for the small additional heat term, namely
mAlcAl(Tr - Tf), since over the small
Tr - Tf interval,
cAl = 0.212 cal/g/K is constant.
T,
-provided that the final temperature of the water, Tf, is room temperature, Tr. It is hard to arrange for Tf to end up exactly at room temperature, but if Tf is close to Tr, one can accurately correct the calorimeter data for the small additional heat term, namely
mAlcAl(Tr - Tf), since over the small
Tr - Tf interval,
cAl = 0.212 cal/g/K is constant.
![\includegraphics[width=4.2in]{figs/fhc2b-3.eps}](img298.gif)
![\includegraphics[width=2.6in]{figs/l103/h04-2.eps}](img301.gif) How much of the N
How much of the N